|
Protons and neutrons are held together by the strong nuclear force, while electrons are held together by the electromagnetic force. How Do Protons, Neutrons and Electrons Interact With One Another? Now that we understand the basics of aluminum’s structure and bonding, let’s take a look at how the particles interact within the atom. Investigating the Structure of Aluminum: Proton, Neutron and Electron Dynamics The number of neutrons does not affect the strength of the bond, but it does affect the stability of the atom. This is because the greater the charge between the atoms, the stronger the attraction will be. The more protons and electrons an atom has, the stronger its bonds will be. The number of protons, neutrons and electrons in an atom affects the strength of its bonds. How Does the Number of Protons, Neutrons and Electrons Affect the Strength of the Bond? Aluminum is a metal, so it forms metallic bonds with other metals and non-metals. Non-metals, on the other hand, do not share electrons and instead form ionic bonds. When metals form bonds, they share electrons in a process known as metallic bonding. What Is the Difference Between Metallic and Non-metallic Bonding? Let’s take a closer look at how they interact to form the element. While protons, neutrons and electrons are all involved in the formation of aluminum, they play different roles. What Role Do Protons, Neutrons and Electrons Play in the Formation of Aluminum? Similarly, when two aluminum atoms form a covalent bond, they create a molecule of aluminum oxide (Al2O3). For example, when two hydrogen atoms form a covalent bond, they create a molecule of water (H2O). 
This creates a new molecule with different properties than the individual atoms. When two atoms form a covalent bond, the electrons in the outer shells of both atoms are shared. How Do Protons, Neutrons and Electrons React to Create New Compounds? In either case, the protons and neutrons of both elements remain unchanged. If the electrons are repelled, they will form an ionic bond. If the electrons are attracted to one another, they will form a covalent bond. When aluminum is exposed to other elements, the electrons in its outer shell interact with the electrons in the other element’s outer shell. What Chemical Reactions Occur When Aluminum Is Exposed to Different Elements? Now that we know how protons, neutrons and electrons combine to form aluminum, let’s take a look at how they interact with other elements to form compounds. 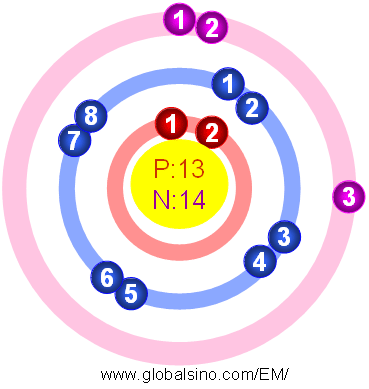
Uncovering the Chemistry of Aluminum: A Look at Protons, Neutrons and Electrons The protons and neutrons also contribute to the mass of the atom, which affects the strength of its bonds. The protons and neutrons are responsible for the element’s identity and stability, while the electrons are responsible for forming chemical bonds. What Is the Role of Each Particle in This Process? This structure gives aluminum its unique properties such as malleability, ductility and electrical conductivity. The atomic structure of aluminum consists of a nucleus containing 13 protons and 14 neutrons surrounded by 13 electrons in three shells. What Is the Atomic Structure of Aluminum? Now that we have a basic understanding of the individual particles that make up aluminum, let’s explore how they combine to form the element. How Do Protons, Neutrons and Electrons Combine to Form Aluminum? Each particle plays an important role in the formation of aluminum and its various properties. Electrons are negatively charged particles that determine the type of bonds an atom forms with other atoms. Neutrons are uncharged particles that provide stability to the nucleus. Protons are positively charged particles that give an atom its identity. What Are the Individual Properties of Each Particle? The protons and neutrons are located in the nucleus of the atom, while the electrons are located in orbitals around the nucleus. This gives aluminum an atomic number of 13 and an atomic mass of 27. How Many Protons, Neutrons and Electrons Does Aluminum Have?Īs previously mentioned, aluminum has 13 protons, 14 neutrons and 13 electrons. Let’s take a closer look at each of these particles. These particles are responsible for the physical and chemical properties of aluminum, as well as its ability to react with other elements. Exploring the Properties of Aluminum: Proton, Neutron and Electron CompositionĪluminum is composed of 13 protons, 14 neutrons and 13 electrons. But what makes aluminum so special? To answer this question, we must explore the atomic structure of the element, including its protons, neutrons and electrons. It has a wide range of applications due to its lightweight, durable, and corrosion-resistant properties. Aluminum is an abundant element found in many everyday objects and materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
